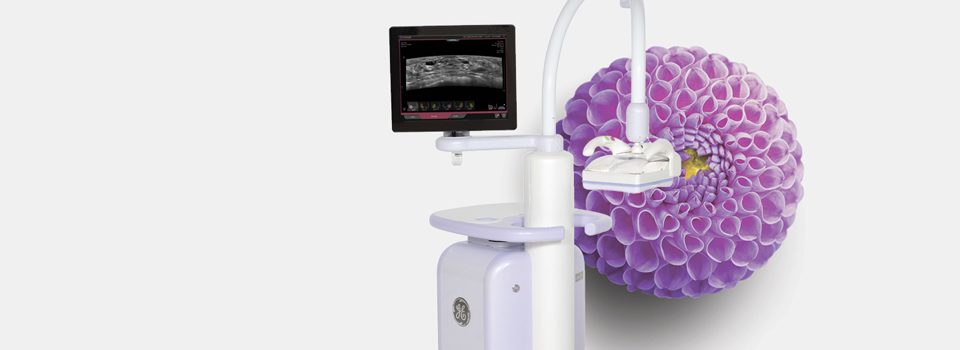
GE INVENIA™ ABUS Automated Breast Ultrasound
Personalized Screening for Patients with Dense Breasts
40% of women have dense breast tissue and for these women, mammography screenings alone may not be enough to find breast cancer. Both cancer and dense tissue appear white on a mammogram, enabling cancer to hide.
Having dense breasts increases a woman’s chances of developing breast cancer by up to six times. In fact, 71% of breast cancers are found in dense breasts.
To help improve early detection, a supplementary screening exam is recommended for dense breasts. Invenia ABUS, Automated Breast Ultrasound, looks differently at dense breast tissue to find cancer that mammography may not see.
Invenia ABUS has been proven to find a 55% relative increase of invasive cancers over mammography alone.
BENEFITS OF ABUS FOR THE PATIENT
- Minimal Compression
- Can breathe normally
- No ionizing radiation
- Non-invasive (no contrast)
- Comfortably lay on back
What makes the Invenia™ ABUS a valuable screening tool?
The Invenia ABUS system is the only FDA ultrasound device approved for breast cancer screening in the United States as an adjunct to mammography for asymptomatic women with dense breast tissue. Invenia ABUS helps physicians differentiate tissue and view tumors more clearly.
The Invenia ABUS system from GE Healthcare is designed to help improve the consistency, reproducibility, and sensitivity of whole breast ultrasound, demonstrating a 35.7 percent improvement in cancer detection (sensitivity) in women with dense breasts without prior breast intervention. (FDA PMA Approval P110006, Sept. 18, 2012.)
Using Invenia ABUS can help physicians provide answers sooner regarding the presence or absence of breast cancer.
Tower Saint John’s Imaging is the premier ABUS Los Angeles service provider, schedule an appointment with us by calling us at 310.264.9000
ABUS Los Angeles, Automated Breast Ultrasound Santa Monica
Tower Saint John’s Imaging
2202 Wilshire Blvd.
Santa Monica, CA 90403
Telephone: 310.264.9000