PLUVICTO is the First and Only Approved PSMA-targeted Radioligand Therapy
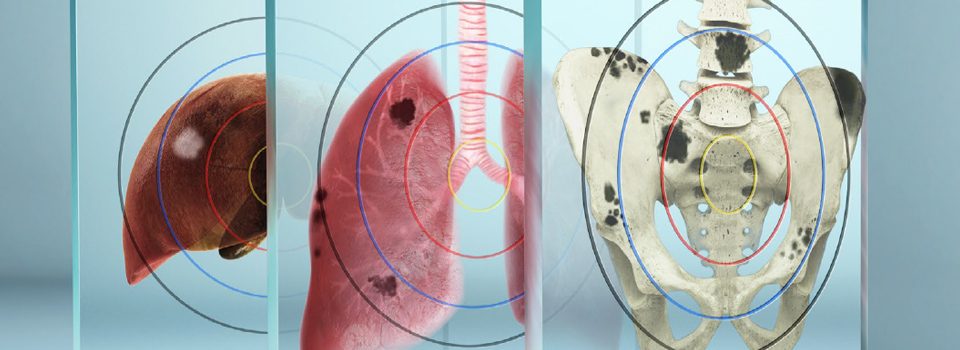
PLUVICTO is a Radiopharmaceutical Used to Treat Adults With Metastatic Castration-resistant Prostate Cancer (mCRPC)
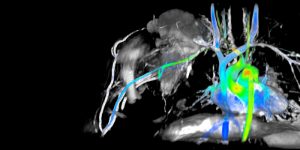
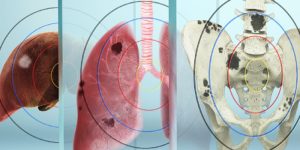
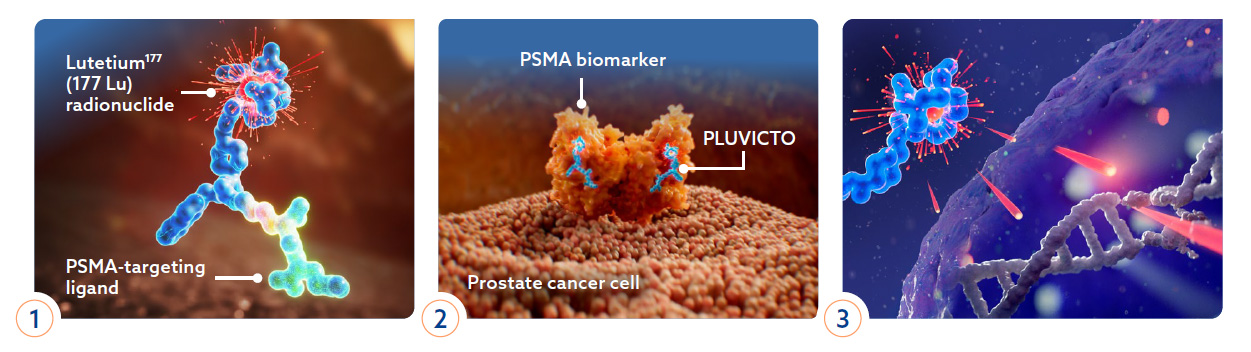
Pluvicto, or Lutetium-177 vipivotide tetraxetan PSMA therapy, is an innovative theranostic solution designed for treating advanced metastatic prostate cancer. This groundbreaking treatment utilizes targeted radiation, aiming specifically at PSMA, a molecule found on the surface of prostate cancer cells. It is complemented by Locametz, also known as gallium-68 gozetotide, a companion diagnostic used in PSMA PET scans to visualize cancer cells expressing PSMA. Approved by the FDA in March 2022, both lutetium-177 PSMA and gallium-68 PSMA have demonstrated significant enhancements in prostate cancer survival rates and patients’ quality of life. Moreover, they have shown efficacy in prolonging the progression-free interval of the disease.
Benefits of Pluvicto in Treating Advanced Metastatic Cancer
- Targeted Treatment: Lutetium-177 PSMA therapy specifically targets prostate cancer cells by focusing on the PSMA molecule, minimizing damage to healthy tissues.
- Improved Diagnosis: Gallium-68 PSMA PET scans provide precise imaging to identify cancerous lesions expressing PSMA, aiding in accurate diagnosis and staging of the disease.
- Enhanced Survival Rates: Clinical studies have shown that these therapies significantly improve survival rates for patients with advanced metastatic prostate cancer.
- Quality of Life: By effectively targeting cancer cells while minimizing side effects, these treatments can enhance patients’ quality of life during and after therapy.
- Disease Progression Delay: Lutetium-177 PSMA therapy and gallium-68 PSMA PET scans have been proven to extend the time it takes for the disease to progress, providing patients with valuable additional time.
- Personalized Medicine: The theranostic approach of combining therapy with companion diagnostics allows for personalized treatment plans tailored to individual patients, optimizing outcomes.
Overall, Lutetium-177 PSMA therapy and gallium-68 PSMA PET scans offer a promising advancement in the management of advanced metastatic prostate cancer, providing both therapeutic benefits and improved diagnostic accuracy.
Potential Side Effects
Lutetium-177 PSMA therapy may lead to fatigue and nausea in certain patients. The radiation can affect the salivary glands, resulting in dry mouth, as these glands, responsible for saliva production, also express PSMA, albeit to a lesser extent. Additionally, blood cell counts are monitored throughout treatment, with occasional temporary dips observed, typically without significant symptoms.
To find out more about Pluvicto in treating adult patients with prostate-specific membrane antigen (PSMA)-positive metastatic castration-resistant prostate cancer (mCRPC) schedule an appointment with us by calling 310.264.9000
For more information visit PLUVICTO® (lutetium Lu 177 vipivotide tetraxetan)
PLUVICTO Treatment for Metastatic Castration-resistant Prostate Cancer Los Angeles, Santa Monica, CA
Tower Saint John’s Imaging
2202 Wilshire Blvd.
Santa Monica, CA 90403
Telephone: 310.264.9000